Name of Lead Author: Solomon Nwaka
Organization: African Network for Drugs and Diagnostics Innovation
Country: Ethiopia
ABSTRACT
Achieving Sustainable Development Goal 3, and related 2030 development goals will require coordination and support for enhanced discovery, development and delivery of essential health technologies within Africa and other developing countries. We present African Network for Drugs and Diagnostics Innovation (ANDI), as a sustainable and bottom-up model to promote research, innovation and access to health technologies to improve health and wellbeing of people developing countries, especially. The model leverages available local capacity to support technology development and market entry to improve health care delivery in Africa. It aims to create a sustainable platform for health innovation in Africa. This means that capacity is built around projects and outcomes and impact are measurable. The core activities of ANDI include: a) Needs and gap analyses as well as identification of viable project opportunities; b) Brokerage and establishment of partnerships among local and external partners (global partnerships) to support project validation, coordination and capacity building as well as transition to scale and market access. Training for management of intellectual property, technology transfer and licensing are integral part of this approach; and c) Advocacy and networking to enhance R&D output through knowledge exchange and open knowledge platforms. We believe that our contribution meets the criteria established by the High Level Panel. The approach creates sustained local innovation, human resource development, public-private partnerships as well as development of private sector and social enterprises in Africa, all in support of increased access to essential health technologies to poor populations. Local regulatory and policy frameworks that support such efforts should be fast-tracked, including through capacity building.
Submission
Promoting Locally Driven and Integrated Model for R&D and Access to Essential Medicines
1. Introduction
In the developing world, over six million people die from neglected diseases such as malaria, sleeping sickness as well as the lesser known parasitic diseases (1). In addition, over 360 million disability-adjusted life years (DALYs) are lost globally due to these diseases (1). The African continent bears the greatest burden of these diseases, and is currently experiencing a surge in deaths due to non-communicable diseases such as cardiovascular disease, cancer, diabetes and respiratory diseases. Unfortunately sustained indigenous research and development (R&D), manufacturing and access mechanisms for health technologies (drugs, vaccines, devices/diagnostics and other health tools) needed by its citizens have not received much attention.
A recent article by heads of the WHO, UNAIDS and UNIDO titled “Commodities for better health in Africa – time to invest locally” (2), states: “Now is the time to forge new partnerships that can support all African countries. The goal is to address health inequities and build capacity to meet supply shortages for essential health commodities that cannot be sourced reliably and sustainably from outside the continent. Africa remains dependent on imported medicines and other health technologies – a risky situation in a continent with the world’s highest prevalence of HIV. The 7.6 million Africans living with HIV now on antiretroviral treatment and millions more waiting depend on 80% of antiretroviral medicines being imported from outside the continent. The local production of medicines and other essential health commodities is important for all health challenges faced by the continent. Demand for health commodities is growing rapidly. The ageing population in Africa requires access to a growing range of medicines and assistive technology that cannot be met with Africa’s existing manufacturing capacity and sources of supply. The world is calling for a new, more inclusive global development framework for the post-2015 era.” While the article focused on local manufacturing, the argument is also true for R&D for health technologies.
1.1 Health R&D and innovation landscape in Africa
A landscape analysis that informed the establishment of the African Network for Drugs and Diagnostics Innovation (ANDI) (3, 4), showed that significant health R&D capacity exists in different parts of Africa, but this capacity is fragmented, uncoordinated, and not properly utilized to address the continent’s health challenges. It further showed that lack of intracontinental collaboration and low levels of investment, contributes to gaps in the continental R&D, a lack of local ownership, and suboptimal utilization of available research capability.
Consistent with these results, a new analysis by ANDI and partners, based on publication outputs by African scientists with their global peers between 2010 and 2015 show the following trends for 20 prevalent African diseases and technologies:
a) Of some 7.5 billion articles produced from 2010 to 2015, only about 0.4% include African authors, suggesting that Africa still lags behind in health research and innovation;
b) A number of African institutions are leaders in their areas of focus, suggesting that scale and technical know-how can be improved if more partnerships are created with established institutions (Figure I);
c) Intra-African networks are not well articulated (Figure I), but national clusters are improving and most of the time a foreign institution serves as a bridge between African countries.
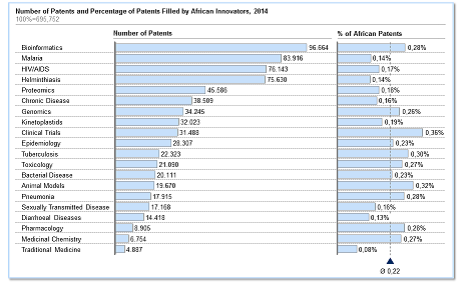
Regarding patents, only about 0.22% of patents filed globally in 2014 for the same indications originated from African innovators, suggesting that Africa represents a small subset in terms of innovation as measured by patent filing, and highlights the need to support local innovation (Figure II).
These data support: a) The need to invest in the translation of African innovations from bench to market by leveraging existing capacity; and, b) The concerted focus of ANDI in building capacity and partnerships to address the health needs of the African population through both intra-African collaboration as well as North-South and South-South collaborations.
Figure I: Pattern of collaboration in traditional medicines R&D. Intra African Collaboration is timid. Cities where most connected institutions are located are shown as top 20 institutions.
Figure II: Patenting trends for key diseases and technologies of interest to Africa. Average of 0.2% of patents emerge from Africa.
We present ANDI as a sustainable and bottom-up model to promote research, development and access to health technologies to improve health and wellbeing, as envisaged by Sustainable Development Goal (SDG) 3, and related 2030 Agenda for Sustainable Development. We discuss the ANDI’s operational model and how it can contribute to the sustainable implementation of SDGs and related global and regional strategies, if properly resourced. Indeed, the work of ANDI touches on others SDGs that deal with innovation, partnerships, poverty alleviation, water and sanitation (5).
2. ANDI Mission, Vision and Operational Model
ANDI’s mission is to promote and sustain African-led health innovation to address the continent’s health needs through the assembly of collaborative networks, and building of capacity to support public health and development.
ANDI’s vision is to create a sustainable platform for health innovation in Africa to address the health challenges of the continent and populations in need.
2.1 Value Proposition
Since 2008, ANDI has been promoting the need to harness the untapped power of local collaboration as well as equitable North-South and South-South partnership, towards health R&D and technology development aimed at solving the basic health challenges of the African continent and other developing countries. It supports translation of local technologies from laboratories to markets. ANDI offers a clear understanding of the medical needs, ongoing health R&D and innovation activities and capabilities, as well as pan-African due diligence on ongoing innovations. Through its initiative on mapping the R&D landscape in Africa, pan-African COEs, Innovation Awards and regional hubs, as well as project identification, fellowships and training, ANDI could determine those R&D programs most likely to lead to potential products that meet the needs of the African population. ANDI’s pan-African granting experience has allowed ANDI access and better understanding of the state of science across more than 50 countries.
2.2 ANDI’s Open Innovation and Business Model
The basic principle for ANDI’s open innovation and business model (Figure III) lies in the realization that a more integrated and holistic partnership approach that shares lessons across diseases and technology platforms promises to be more successful in the African context, where resources are very limited (6).
Figure III: ANDI Innovation and Business Model – aligns available R&D and technology development expertise with specific diseases or health conditions in an integrated and holistic way.
The model leverages available local capacity to support technology development and market entry to improve health care delivery in Africa. This means that capacity is built around projects and outcomes and impact are measurable. The core function and activities of ANDI are: a) Needs and gap analyses as well as identification of viable project opportunities; b) Brokerage and establishment of partnerships among local and external partners (global partnerships) to support project validation, coordination and capacity building as well as transition to scale and market access. Training for management of intellectual property, technology transfer and licensing are integral part of this approach; and c) Advocacy and networking to enhance R&D output through knowledge exchange and open knowledge platforms.
2.3 ANDI as a Facilitator and Catalyst
ANDI’s Strategic Plan defines the role of ANDI as a “broker”, “facilitator” and “catalyst” in advancing local technologies with potential for impact in Africa and other developing countries (6). To this end, ANDI is focusing on filling a critical gap by driving local innovations out of the so-called 'valley of death' that will lead to eventual commercialization and impact. This will not only have positive impact on public health, but also human resource development and job creation - all of which contribute to sustainable development. In the developed world and advanced economies, clear processes for addressing resource needs that bring concepts into development exist, but in developing countries especially within Africa, such mechanisms are not readily available, and where they do, they are in their infancy.
Consistent with the above, recent reports have shown that numerous technologies that come out of African laboratories with the potential to support healthcare are not further developed due to lack of suitable mechanisms to support rigorous proof of concept, scale up, commercialization and marketing (7, 8, 9, 10). Also, local mechanisms for addressing transition of technologies from laboratory to industry, including IP and financing mechanism are not well established.
These are significant gaps and a niche that ANDI is focusing on through partnership with others. The ANDI-supported bCPAP (Pumani) for newborn with respiratory distress and phototherapy device for children with jaundice, progression of diagnostic kits to support the control and elimination of schistosomiasis, cell phone based coding system for diagnosis and monitoring of disease, and the reformulation of NIPRISAN for sickle cell disease in Africa, are such projects which require a truly innovative financing model to further support validation, industry scale up and market access. Figure IV shows a summary of ANDI’s growing portfolio of project and initiatives
Figure IV: ANDI Portfolio – specific projects are circled. 43 ANDI pan African Centres of Excellence are shown in blue. These centres support project and capacity building. ANDI’s innovation awards and needs assessment initiatives are not shown.
The implementation of these activities is supported by a growing number of pan-African Centres of Excellence (CoEs), which ANDI identifies, based on a transparent and criteria based process. These CoEs are the engine for project implementation, capacity building, tech-transfer and concerted North-South and South-South collaborations.
3. Lessons Learned, Challenges and Opportunities
ANDI has passed through several challenges and successes, and as a growing organization has drawn lessons to improve its work.
Lessons Learned: Some of the lessons include: a) Many African institutions focus on R&D that can address local health needs but lack sustained support to translate research into policy and health products. Given adequate incentives, African institutions are very keen on working together or partnering on projects that can transform health care delivery in Africa; b) Intra-African collaborations or networks as well as local leadership are very important for success; c) The perception by many from within and outside of Africa is that ANDI’s vision, mission and modus operandi are unique and more relevant today than ever before; and, d) The ANDI model can make significant impact with modest but steady funding stream (which is presently lacking).
Challenges: Human and financial sustainability remains a major challenge for ANDI and its work. Experiences over the past few years have shown that ANDI can make significant contribution to enhancing health innovation in Africa with a modest but steady funding stream. ANDI is working on two potential financing or resource mobilization pathways to support its work and attain sustainability: a) Grant and subscription-based funding for the generation of core and non-core funding to be held as Pooled Fund; and, b) An Impact or Social Venture Fund, to be professionally managed for growth and impact.
ANDI’s human resource challenges are linked to financing. Presently, ANDI has nine full time equivalents in addition to successful use of volunteers secured through the United Nations Volunteers program. Additionally, ANDI has established as secondment program whereby partners and stakeholders can second staff to the ANDI Secretariat. While these approaches are promising and should be promoted more broadly, ANDI needs a critical mass of full time Secretariat staff to support its technical, advocacy and fundraising work – this will contribute to sustainability.
Opportunities: Notwithstanding the challenges described above, some of the opportunities that ANDI is working to maximize include: a) Post-2015 development agenda and SDGs as well as relevant regional declarations in Africa such as the AU Agenda 2063; b) Increasing interest to support and advance local innovations, through regional R&D, manufacturing and regulatory initiatives; c) Limited pan-African initiatives engaged in the implementation of global and regional strategies or resolutions on health R&D and technology development; d) Expressed commitments by stakeholders including African government and development partners in support of initiatives like ANDI, such as the Global Strategy and Plan of Action (GSPoA) on Public Health, Innovation and Intellectual Property (10), various G7 and regional commitments; e) Over 40 CoEs spread across the continent with potential for collaboration on projects and capacity building; g) Enthusiasm by African researchers, institutions and CoEs to become more engaged in addressing the needs of the continent; and f) Need to validate the regional regulatory harmonization efforts in Africa with projects that emerge from Africa (11), for example through the work of ANDI and its partners.
4. Organizational Structure
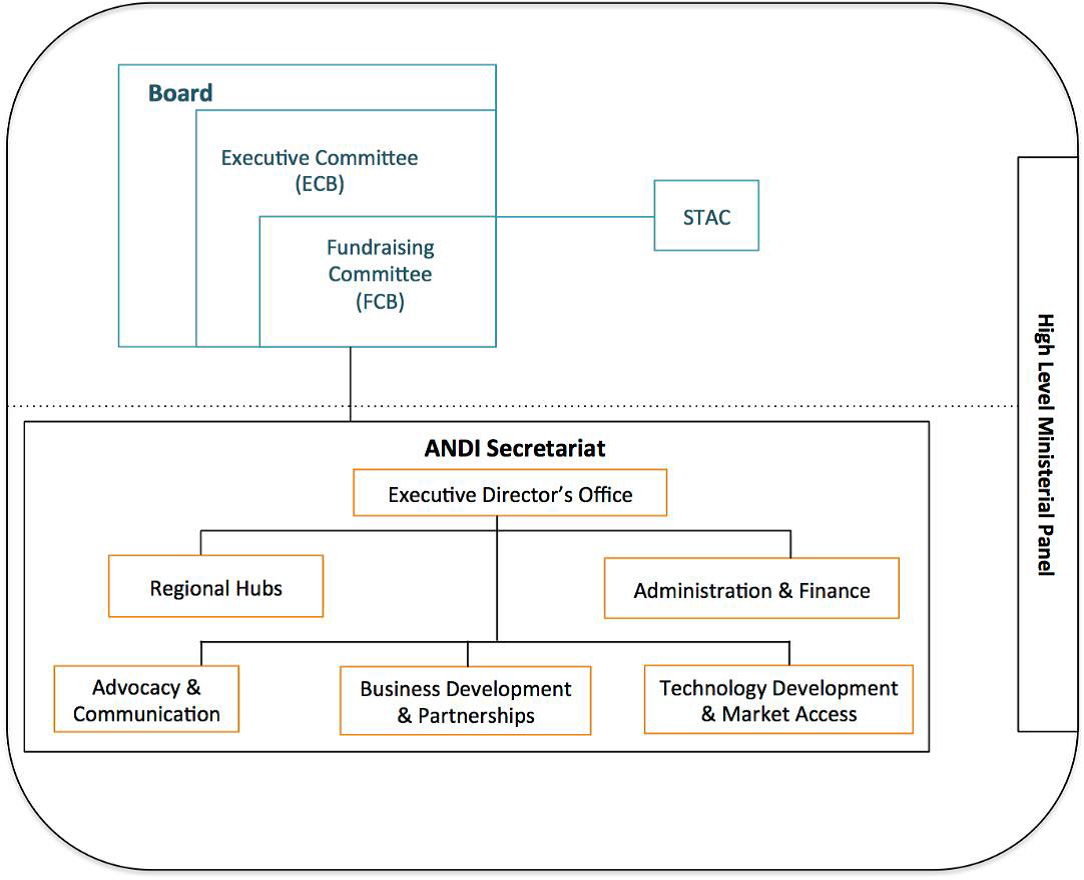
ANDI’s organizational structure consists of the governing bodies and Secretariat (Figure V). This structure acknowledges the substantial geographical, cultural, and epidemiological differences within the Continent as well as the need for robust governance and oversight support structures.
Figure V. ANDI Organization Structure
4.1.1. ANDI Governance
The Board is the highest governing body that ensures a wider stakeholder representation. It drives strategic design and policy making, which in turn are implemented and operationalized by the Secretariat. The responsibilities of the Board are three-fold: a) Determines ANDI’s strategy and priorities; b) Approves work-plans and budget; and c) Defines the advocacy strategy and supports its implementation by engaging with governments, businesses and individuals.
The three committees of the Board include: a) The Executive Committee of the Board (ECB) with the responsibility of regularly monitoring the work of the Secretariat and making recommendations to the Board. The ECB also provides policy and operational support to the ANDI Secretariat. The ECB is made up of the Chairperson, Vice Chair and Executive Director of ANDI; b) Fundraising Committee of the Board (FCB) that supports the Board and Secretariat on fundraising in accordance with its terms of reference. Its membership includes the Chair of the Board, Executive Director, and other representatives approved by the Board; and, c) The Scientific and Technical Advisory Committee (STAC) that provides independent scientific and technical advice to the Board and the ANDI Executive Director in accordance with its terms of reference. Although STAC is highlighted as a committee of the Board, it should be noted that STAC is an independent body that is composed of independent experts acting on their individual capacity.
4.1.2. ANDI Secretariat
ANDI is a partnership presently hosted by and derives its legal status from the United Nations Office of Project Services (UNOPS). That said, the long-term goal of ANDI is to become a fully independent intergovernmental entity. A fully functional ANDI Secretariat will continue to be lean (about 22 full time Secretariat staff plus volunteers), with a headquarters or central office and regional hubs in the five regions of Africa (Figure VI). Presently, ANDI is working with about nine full time staff members and 4 volunteers.
Figure VI: ANDI Secretariat Organogram. Note the lean and relatively flat structure of the ANDI Secretariat.
5. Monitoring, Evaluation and Reporting
A results based monitoring and evaluation framework with inputs, outputs, outcomes and impact are employed by the ANDI. Clear indicators that guide the Secretariat in its daily work as well as support the ANDI Board, STAC and Stakeholders in their oversight and evaluation of the work of ANDI. Risk assessment and management practices are employed in the management and oversight of ANDI.
6. Conclusion
We believe that our contribution meets the criteria established by the High Level Panel: a) it addresses policy incoherence as they relate to inequity in innovation and access to medicines, and promotes use of existing international frameworks such as TRIPS flexibilities. Our approach delinks the cost of R&D from the price of final product, b) the outcomes and impact of our approach include sustained access to easy to use, affordable and quality health technologies. The approach create sustained innovation, human resource development, public-private partnerships as well as development of private sector and social enterprises in Africa in support of local innovation, c) Our contribution requires enhanced political and financial support for the goals to be achieved and sustained. The institutional and governance structures are in place and there is room to further strengthen them. Our contribution advances sustainable health outcomes by ensuring that that the people most affected by diseases take the lead in addressing the health challenges that affect them the most, and d) Our contribution and approach is evidence based.
Imagine a world where African countries, initiatives and laboratories are contributing significantly in the discovery, development and delivery of the health products that they need the most. Local regulatory and policy frameworks that support such efforts should be fast-tracked, including through capacity building. A successful implementation of the SDGs would require such a bottom up approach.
References and Bibliography
1. Institute for Health Metrics and Evaluation (IHME). Global Burden of Disease Study 2010 (GBD 2010) Results by Cause and by Region 1990-2010, 2013. [Accessed online Feb 9th 2016]: http://ghdx.healthmetricsandevaluation.org/global-burden-disease-study-2010-gbd-2010-data-downloads
2. Sidibé M, Yong L, Chan M. Commodities for better health in Africa – time to invest locally, 2014. Bulletin of the World Health Organization; 92:387-387A. DOI: http://dx.doi.org/10.2471/BLT.14.140566
3. The Strategic and Business Plan for ANDI 2010-2015. [Accessed online Feb 9th 2016]: http://www.who.int/tdr/publications/documents/sbp_andi.pdf
4. Nwaka S. et al. Developing ANDI: A Novel Approach to Health Product R&D in Africa, 2010. PLoS Med 7(6): e1000293. DOI: 10.1371/journal.pmed.1000293. http://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1000293
5. Sustainable Development Goals. [Accessed online Feb 9th 2016]: http://www.un.org/sustainabledevelopment/
6. The Strategic Plan for ANDI 2016 – 2020 (to be publicized shortly).
7. Nwaka S. et al. Analysis of pan-African Centres of excellence in health innovation highlights opportunities and challenges for local innovation and financing in the continent, 2012. BMC Int Health Hum Rights 12(11): DOI: 10.1186/1472-698X-12-11.
8. Nordling L. Changing council, 2014. Nature Medicine 20:113–116 DOI:10.1038/nm0214-113.
9. Mackintosh (Ed) et al. Making Medicines in Africa; The Political Economy of Industrializing for Local Health 2015. [Accessed online Feb 9th 2016]: http://www.palgraveconnect.com/pc/doifinder/view/10.1057/9781137546470
10. The Global Strategy on Public Health Innovation and Intellectual Property. [Accessed online Feb 9th 2016]: http://www.who.int/phi/publications/gspa-phi/en/
11. Ndomondo-Sigonda M, Ambali A. The African Medicines Regulatory Harmonization Initiative: Rationale and Benefits, 2011. Clin Pharmacol Ther. DOI: 10.1038/clpt.2010.299